Top Chemicals Used in Electroplating for Enhanced Performance and Durability?
Electroplating is a vital process in many industries, enhancing the performance and durability of products. It involves the application of a metallic coating to substrates to prevent corrosion and wear. As highlighted in a report by the Electroplating Association, the global electroplating market is projected to reach $14 billion by 2025. This growth emphasizes the importance of understanding the chemicals used in electroplating.
Dr. Emily Hart, a leading expert in surface finishing, states, "The right chemicals can significantly improve the lifespan of plated components." In electroplating, chemicals such as nickel sulfate, copper sulfate, and hexavalent chromium play crucial roles. Each of these chemicals affects the quality and performance of the electroplated layer. However, mismanagement of these chemicals can lead to undesirable results, like poor adhesion or uneven plating.
Despite advancements, challenges remain in the industry. Not all manufacturers fully grasp the sensitivity of chemical mixtures. Some continue to rely on outdated practices, risking product integrity. The balance of using innovative chemicals while maintaining safety is a constant consideration in electroplating. Understanding these dynamics is essential for achieving optimal results in the electroplating process.

Top Chemicals in Electroplating: An Overview of Key Materials
Electroplating is a crucial process in various industries, enhancing the durability and performance of metal surfaces. The choice of chemicals plays a significant role in achieving desired outcomes. Commonly used chemicals include nickel sulfate, chromium trioxide, and copper sulfate. According to recent industry reports, nickel sulfate accounts for approximately 35% of the global electroplating market. Its properties contribute to improved hardness and corrosion resistance.
Another essential compound is chromium trioxide, known for creating a bright and durable finish. Its importance is reflected in its increasing application in automotive and aerospace industries. However, it's important to note potential environmental concerns related to chromium compounds. Companies need to approach this with caution and consider safer alternatives when possible.
Tip: Always assess the environmental impact of the chemicals you use. Look into cleaner and greener alternatives that might fulfill the same role.
Copper sulfate is widely used, especially in decorative plating. It provides excellent conductivity and a bright appearance. Despite its benefits, users should evaluate its long-term effects and be ready to adapt their processes. Investing in research can lead to better sustainability.
Tip: Regularly review your chemical supply chain. Stay informed on new innovations that can help enhance performance while reducing negative impacts.
Top Chemicals Used in Electroplating for Enhanced Performance and Durability
| Chemical Name | Function | Application | Properties |
|---|---|---|---|
| Nickel Sulfate | Provides a nickel source for electroplating | Used in decorative and functional nickel plating | Corrosion-resistant, can enhance hardness |
| Copper Sulfate | Acts as a primary source of copper for plating | Used in electronic components and decorative items | Excellent conductivity, improved surface finish |
| Chromic Acid | Provides a source of chromium for plating | Used in hard chrome plating for industrial applications | High hardness, good wear resistance |
| Zinc Chloride | Acts as an electrolyte in zinc plating | Used in corrosion protection applications | Good corrosion resistance, cost-effective |
| Tin Fluoride | Enhances the quality of tin deposits | Used in food packaging and electronics | Improved adhesion, better solderability |
Role of Electrolytes in Enhancing Electroplating Performance

Electrolytes play a critical role in electroplating processes. They determine the quality and consistency of the plating layer. A well-chosen electrolyte enhances the adhesion between the substrate and the metal layer. This adhesion prevents peeling and increases durability in various applications.
Commonly used electrolytes include sulfates, chlorides, and phosphates. Each has distinct properties that can improve conductivity and overall plating efficiency. For instance, sulfates can provide a smooth finish, while chlorides tend to encourage faster plating rates. Yet, not every electrolyte suits every metal. That mismatch can lead to issues.
Often, the pH level of the electrolyte is overlooked but it significantly influences the electroplating outcome. A slightly acidic or basic solution can alter plating quality. It’s essential to test and adjust the pH regularly to achieve optimal results. When errors occur, they often stem from ignoring these details. Adjustments and careful monitoring are critical in achieving excellent plating performance.
Common Metal Deposits: Nickel, Copper, and Chromium Analysis
Electroplating is crucial for enhancing metal durability. It provides a thin metallic coating that improves corrosion resistance and wear. Let's focus on three common deposits: nickel, copper, and chromium. Each metal offers unique benefits and challenges.
Nickel is popular for its toughness and lustrous finish. It creates a barrier against moisture and oxidation. However, the process can be difficult to control. Variations in thickness may lead to uneven surfaces. Copper, on the other hand, is excellent for conductivity. It's often used as an undercoat. Yet, it can tarnish quickly, requiring additional protective layers. This adds complexity to the process.
Chromium deposits are known for their hard, shiny surface. They create an aesthetically pleasing exterior. But achieving a uniform coating with chromium can be a challenge. There are health concerns associated with its use as well. Understanding these metals helps in making informed decisions. There’s a need for constant evaluation and refinement in electroplating methods for better results.
Environmental Impact and Safety Regulations in Electroplating Chemicals
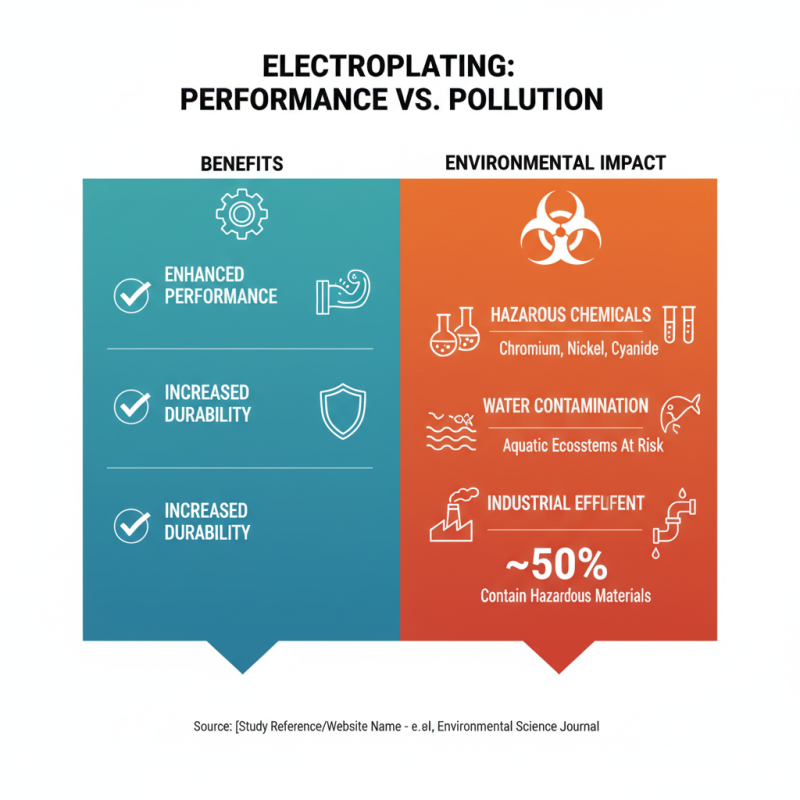
Electroplating is widely praised for enhancing performance and durability of metal components. However, the chemicals used in this process have significant environmental impacts. Heavy metals like chromium and nickel are common in electroplating solutions. These substances can contaminate water sources, posing risks to aquatic ecosystems. A study noted that approximately 50% of industrial effluents in electroplating contain hazardous materials.
Safety regulations are crucial in mitigating these risks. The EPA enforces strict limits on metal discharges. Facilities must adopt proper waste management practices to comply. This includes treating effluent before release and recycling chemicals wherever possible. Maintaining a safe working environment is also essential. Protective gear must be worn to prevent exposure to toxic materials.
Tips: Regularly review safety guidelines. Invest in training for staff to handle hazardous materials. Consider adopting greener alternatives for electroplating processes. Emphasizing sustainability not only protects the environment but can also enhance the company’s reputation. Seeking third-party certifications can help ensure adherence to safety regulations.
Innovations in Electroplating Chemicals for Improved Durability
In recent years, the electroplating industry has seen significant innovations in the chemicals used for enhanced performance and durability. Advanced formulations are now designed to create thinner, more resilient coatings. According to a report by the Electroplating Association, the market for specialty plating chemicals is expected to grow by 4.5% annually, driven by demand for high-performance coatings in automotive and electronics sectors.
One promising area of development is the use of eco-friendly chemicals. Safer alternatives to traditional cyanide-based solutions have begun to emerge. These newer solutions not only reduce environmental impact but also enhance substrate adhesion. Additionally, some studies indicate that these innovations can increase corrosion resistance by up to 30% compared to conventional methods.
However, challenges remain. The transition to new chemical formulations may lead to initial compatibility issues with existing processes. Some industries may need to adapt their equipment to fully leverage these innovations. It’s crucial to conduct further research on the long-term performance of these new chemicals under varying conditions. The evolving landscape of electroplating chemicals presents both opportunities and hurdles that require careful consideration and ongoing evaluation.
Related Posts
-

2025 Top Guide to Cobalt Plating Techniques and Applications
-

How to Choose the Best Industrial Hard Chrome Plating Process for Your Needs
-

What is Decorative Plating and How Does it Enhance Product Aesthetics
-

Top 10 Tips for Successful DIY Chrome Plating at Home
-

How is Chromium Used in Electroplating? Advantages and Applications Explained
-

2026 Best Industrial Chrome Plating Techniques for Enhanced Durability?
